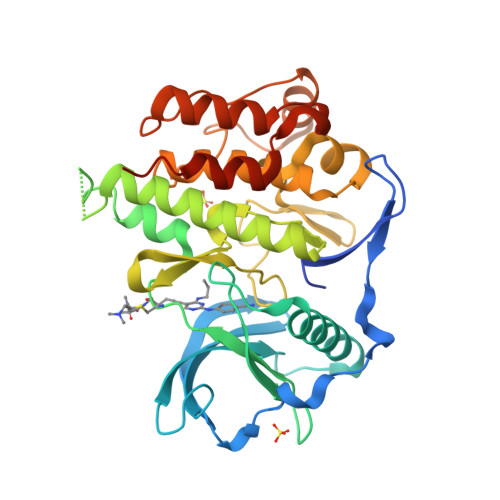
A novel irreversible FLT3 inhibitor, FF-10101, shows excellent efficacy against AML cells withFLT3mutations.
Yamaura, T., Nakatani, T., Uda, K., Ogura, H., Shin, W., Kurokawa, N., Saito, K., Fujikawa, N., Date, T., Takasaki, M., Terada, D., Hirai, A., Akashi, A., Chen, F., Adachi, Y., Ishikawa, Y., Hayakawa, F., Hagiwara, S., Naoe, T., Kiyoi, H.(2018) Blood 131: 426-438
- PubMed: 29187377
- DOI: https://doi.org/10.1182/blood-2017-05-786657
- Primary Citation of Related Structures:
5X02 - PubMed Abstract:
An activating mutation of Fms-like tyrosine kinase 3 ( FLT3 ) is the most frequent genetic alteration associated with poor prognosis in acute myeloid leukemia (AML). Although many FLT3 inhibitors have been clinically developed, no first-generation inhibitors have demonstrated clinical efficacy by monotherapy, due to poor pharmacokinetics or unfavorable safety profiles possibly associated with low selectivity against FLT3 kinase. Recently, a selective FLT3 inhibitor, quizartinib, demonstrated favorable outcomes in clinical studies. However, several resistant mutations emerged during the disease progression. To overcome these problems, we developed a novel FLT3 inhibitor, FF-10101, designed to possess selective and irreversible FLT3 inhibition. The co-crystal structure of FLT3 protein bound to FF-10101 revealed the formation of a covalent bond between FF-10101 and the cysteine residue at 695 of FLT3. The unique binding brought high selectivity and inhibitory activity against FLT3 kinase. FF-10101 showed potent growth inhibitory effects on human AML cell lines harboring FLT3 internal tandem duplication ( FLT3 -ITD), MOLM-13, MOLM-14, and MV4-11, and all tested types of mutant FLT3-expressing 32D cells including quizartinib-resistant mutations at D835, Y842, and F691 residues in the FLT3 kinase domain. In mouse subcutaneous implantation models, orally administered FF-10101 showed significant growth inhibitory effect on FLT3-ITD-D835Y- and FLT3-ITD-F691L-expressing 32D cells. Furthermore, FF-10101 potently inhibited growth of primary AML cells harboring either FLT3 -ITD or FLT3 -D835 mutation in vitro and in vivo. These results indicate that FF-10101 is a promising agent for the treatment of patients with AML with FLT3 mutations, including the activation loop mutations clinically identified as quizartinib-resistant mutations.
Organizational Affiliation:
Pharmaceutical and Healthcare Research Laboratories, FUJIFILM Corporation, Kanagawa, Japan.